How to Switch to eQMS Software When You're Just Using Paper

How reliable is your paper-based quality management system?
This situation plays out in medical device companies everywhere:
There was a situation when I was consulting for a company when the FDA showed up to conduct a routine inspection.
FDA asked the client for their CAPA log, which they’d filled out showing which ones were completed and which were still in process. FDA put a star next to a couple CAPA on the list that they wanted to look into further, and a "runner" was dispatched to find the relevant files.
Bottom line, they couldn’t find one of the CAPAs. At all.
They had to come back and say “we don’t know where it is”, which is not a great sign for the FDA. This became a 483 observation, although about a week later they were able to locate it, buried in a desk drawer.
This is just one example of several horror stories I have seen related to paper-based systems and the tendency for important documents to get misplaced or completely lost. No one wants to receive a FDA 483 observation or audit observation, but this is what will result if you can’t provide the evidence being requested.
Non-compliance is the single biggest risk facing your medical device company. Are you going to trust it to paper?
Free Bonus Giveaway: Are you ready for an eQMS? Here are 7 signs to know if you are.
Why paper-based medical device companies are at risk
This story is in no way the first of its kind nor will it be the last. Sure, some companies have managed fairly well with paper-based systems, but that doesn’t negate how open they are to human error. It doesn’t take much for a piece of paper to get lost or for key decision-makers to fall out of the loop due to errors in paperwork.
When these things happen, your company can be put at risk. What happens if a project gets shut down due to missing compliance documents? Can your company afford a drawn-out time to market because you need to spend time on inspection and audit observations or updating documentation?
6 problems I regularly see with paper-based QMS tools:
- Missing documents. As highlighted in my opening story, missing documents can lead to an unsatisfactory FDA inspection and/or ISO audit.
- People like to use Post-It notes, especially in the design history file. Often these don’t necessarily apply to the document, leading to confusion, or they’re used as a reminder and whatever the situation was never got addressed. Post-It notes can signify red flags to inspectors because why are they there if all documents are up-to-date? (Note, don't use Post-It notes in your DHF. This is a bad idea and poor practice!)
- What tends to happen to documents as they get passed around? Often, each person might make changes or mark-ups as they receive the document. This means that key people don’t have overall visibility where they’ve already signed off on the doc. This is a terrible practice, as your decision-makers think they’ve signed off on something when in fact changes have been made since that they don't even know about.
- Missing signatures. I have experienced this to his chagrin as well. Imagine confidently passing some paperwork over to an FDA inspector thinking that everything was complete, then the first thing they turn to is the signature page which, you guessed it, has missing signatures…
- Communicating with remote teams and suppliers is difficult. The trend for offices and teams spread across the globe has only grown so that it’s becoming rarer to find everyone co-located. Paper ends up being shipped from one location to the next. Email might be used for document sharing, but this creates a different problem because keeping tabs on the most up to date version that has been tossed around via email is challenging, at best. What happens for example, when someone marks up changes?
- Sometimes companies think of the paper as the “master copy”, even though it is more likely that electronic copies stored on local hard drives or your server is different. Sometimes no one is looking at that original paper document and it may be well out-of-date.
How are you going to make sure you can find a document? Track revisions? Update them? Get signatures? Work with virtual employees? Who’s even using them?
The solution is pretty simple, but one that so many medical device companies seem averse to. For these device makers, it’s time to make the switch from paper to eQMS software.
What is eQMS software?
Greenlight Guru's eQMS software is “the simplest way for your team to manage workflows, reduce risk and improve time to market.”
We created Greenlight Guru specifically for the unique needs of medical device development. Unlike general-purpose QMS tools, Greenlight Guru has been designed to align 1 to 1 with medical device regulations like ISO 13485:2016 and FDA 21 CFR Part 820 immediately out of the box without customization.
The documents and records you generate as part of bringing a new medical device to market are necessary for your team. This documentation establishes sound, repeatable processes for you to measure and analyze in order to ensure maximum efficiency. Managing with intention is important - why would you trust that to the fallible nature of paper?
An eQMS for medical device companies need to have few key aspects:
-
It has pre-built workflows to match regulations and automatically create documents and records.
-
It aligns your processes meet all key regulations.
-
It provides documented evidence that you’re following procedures.
-
It is designed for FDA 21 CFR Part 11 compliance and provides full detailed activity history.
An eQMS skips the cumbersome nature of paper-based systems by creating a single source of truth. Everything is kept in one place, is easily accessible and is kept up-to-date without some scanned, marked-up version floating around elsewhere. Version control is automatic. It’s obvious who has reviewed, approved an item and what the status is.
Greenlight Guru is purpose-built for medical device companies and so includes automated workflows and document generation based on the requirements unique to medical device development. You can share up-to-date information across the world easily.
Click here to learn more about Greenlight Guru's Quality Management Software
How to transfer to eQMS Software
Let’s say you’re a paper-reliant business right now. Your processes are maintained in a labyrinthine filing system, the methodology of which is so arcane it’s only truly understood by one person who left the company 20 years ago.
The first question I often ask is, “are you happy with your QMS as it is?” Sometimes the company is (some people do okay with paper), but it’s about scaling and efficiency. Greenlight Guru’s customer success team can help to build a quality plan and look at when, how and if to transition to eQMS. It’s a collaborative effort between Greenlight Guru and the company.
Sometimes a company really wants a full overhaul because they know the system doesn’t work (often the hard way). Either way, we’re going to help with a plan to get there. It’s not difficult to make the shift.
Typically, our competitors take 6 months to a year to fully implement. Greenlight Guru is much quicker. We believe you should start seeing value ASAP, so we have designed Greenlight Guru so that it’s ready to go out of the box. You can start using Greenlight Guru on day 1.
We’re purpose-built for the medical device industry with those regulations in mind, as opposed to competitors who may be more generalized. We can work with you to have all legacy information transferred within a short time, typically under 2 months--often much faster.
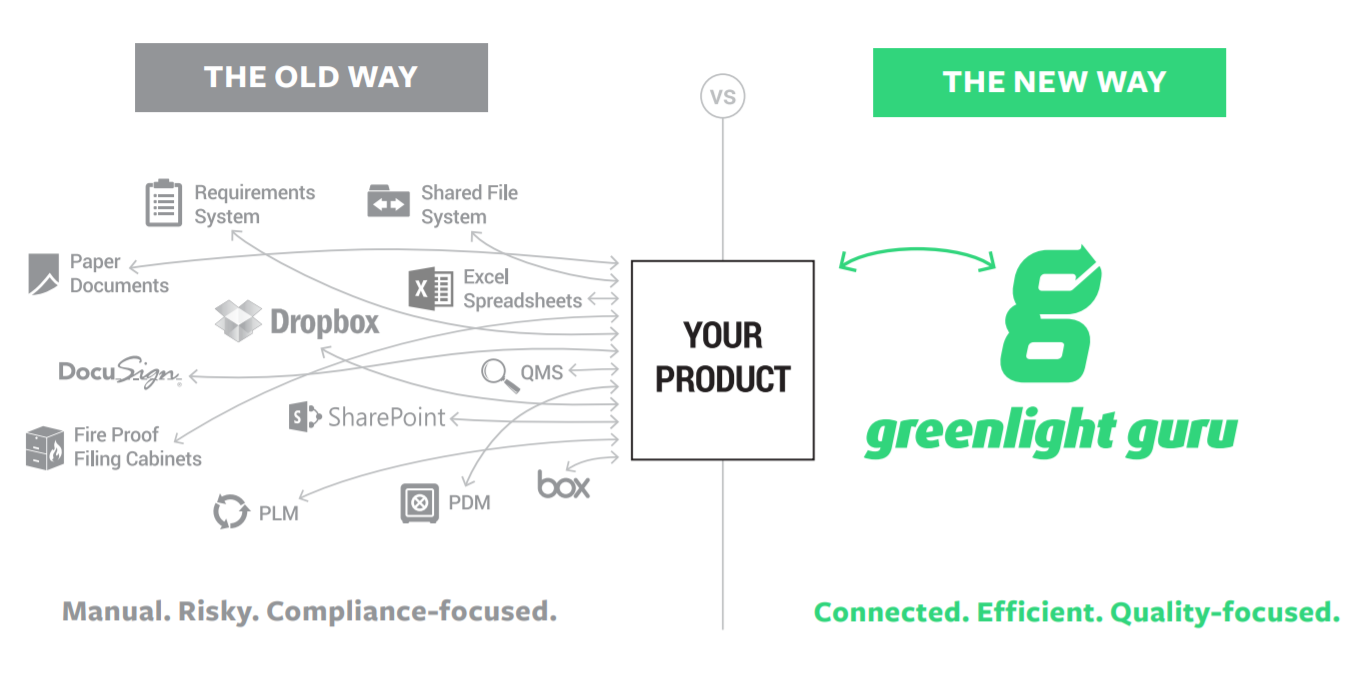
Old way vs. new way
“Market leaders combine people, processes and technology capabilities to create an environment of closed-loop quality.” - LNS Research
The old way was a conglomeration of file cabinets, server folders or paperwork organized by functional area. People had information stored all over the place or used general-purpose tools, like Google Drive, with no integration to tie everything together.
The new way is the Greenlight Guru platform as it ties everything together. Document management is the key layer of the system to capture and prove everything that you have done. It’s about connecting the dots and providing a single source of truth.
For example let’s look at a complaint. Under an old system, you open a complaint investigation and you try to figure out whether anyone else has called with the same problem. You search lists and databases looking for things that might fit.
You also need to determine if you’ve ever done work, like a CAPA on the same thing before. Again, more searching. You take action on the complaint and now you have to feed it back and ensure everything ties together, including design control elements and any other system impacted.
Greenlight Guru interfaces all of those things together. For example, keyword searches help you to identify information in a few clicks. Traditionally you’d have those automations running from an on-site server that links to your local network like this:
As you can see, there are multiple points where inefficiencies may occur. When you leverage a cloud-based system like Greenlight Guru, you have the benefit of automated workflows, which ensures that key items are updated together.
Free Bonus Giveaway: Are you ready for an eQMS? Here are 7 signs to know if you are.
Time to make the switch to eQMS software?
It just makes sense to use technological advances for creating scalability and efficiency in your business. Leading medical device development companies are able to get products to market more quickly and reduce errors by having a robust enterprise quality management software.
Jon Speer is a medical device expert with over 20 years of industry experience. Jon knows the best medical device companies in the world use quality as an accelerator. That's why he created Greenlight Guru to help companies move beyond compliance to True Quality.
Related Posts
[VIDEO] 4 Medical Device Manufacturing Best Practices for a Successful Design Transfer (Approve Phase)
Scaling Regulatory and Quality with Your Company
[VIDEO] 7 Ways to Track QMS Effectiveness using Medical Device Post-market Quality Processes (Track Phase)
Get your free resource
7 Signs you're ready for an eQMS