Best QMS software for medical devices: comparison guide

Choosing the best QMS software for medical devices is one of the most consequential infrastructure decisions a MedTech company makes. The challenge is not finding options. It's knowing how to compare QMS software based on the workflows, regulatory requirements, traceability needs, and quality processes that actually matter for medical device teams.
Most teams approach QMS selection as a compliance checkbox: what is the minimum system that keeps us out of trouble? That framing often leads to underinvestment early and expensive remediation later. A better question is: which medical device QMS software will help your team build compliant, submission-ready records without adding administrative drag to engineering, quality, and regulatory teams?
This guide breaks down the five categories of QMS solutions available to medical device companies today, explains what each one actually costs in practice, and helps you understand which type of system fits where you are in your development journey.
Table of Contents
What to look for in a QMS: the evaluation criteria that actually matter
Comparing the best QMS solutions for medical devices
Best QMS software for different medtech companies
How to evaluate your options: questions worth asking in every demo
Who This QMS Software Comparison Is For
This guide is for medical device teams evaluating QMS software for building a first compliant QMS before submission, moving from paper or spreadsheets to an eQMS, or choosing a system that can scale from pre-market development through commercialization.
How to choose QMS software for medical device companies
Regulatory alignment out of the box
Any QMS you use to manage quality records needs to reflect the current regulatory framework under the FDA's QMSR and ISO 13485:2016. The system you implement should reflect current requirements from day one, not require you to build regulatory alignment yourself through configuration.
Pre-validated workflows
Any software used to manage quality records is subject to computer system validation (CSV) requirements. That validation burden varies significantly depending on the type of system you choose.
Bidirectional traceability
A QMS needs to maintain live, bidirectional traceability between user needs, design inputs, outputs, verification and validation, and risk records.
Implementation speed
A 510(k) typically takes 18 to 36 months from working prototype to cleared device. Understand the realistic timeline from kickoff to a system your team is actually working in before you commit.
Scalability without a platform switch
The system you choose now should be capable of supporting your quality needs through submission and into post-market, not just where you are today.
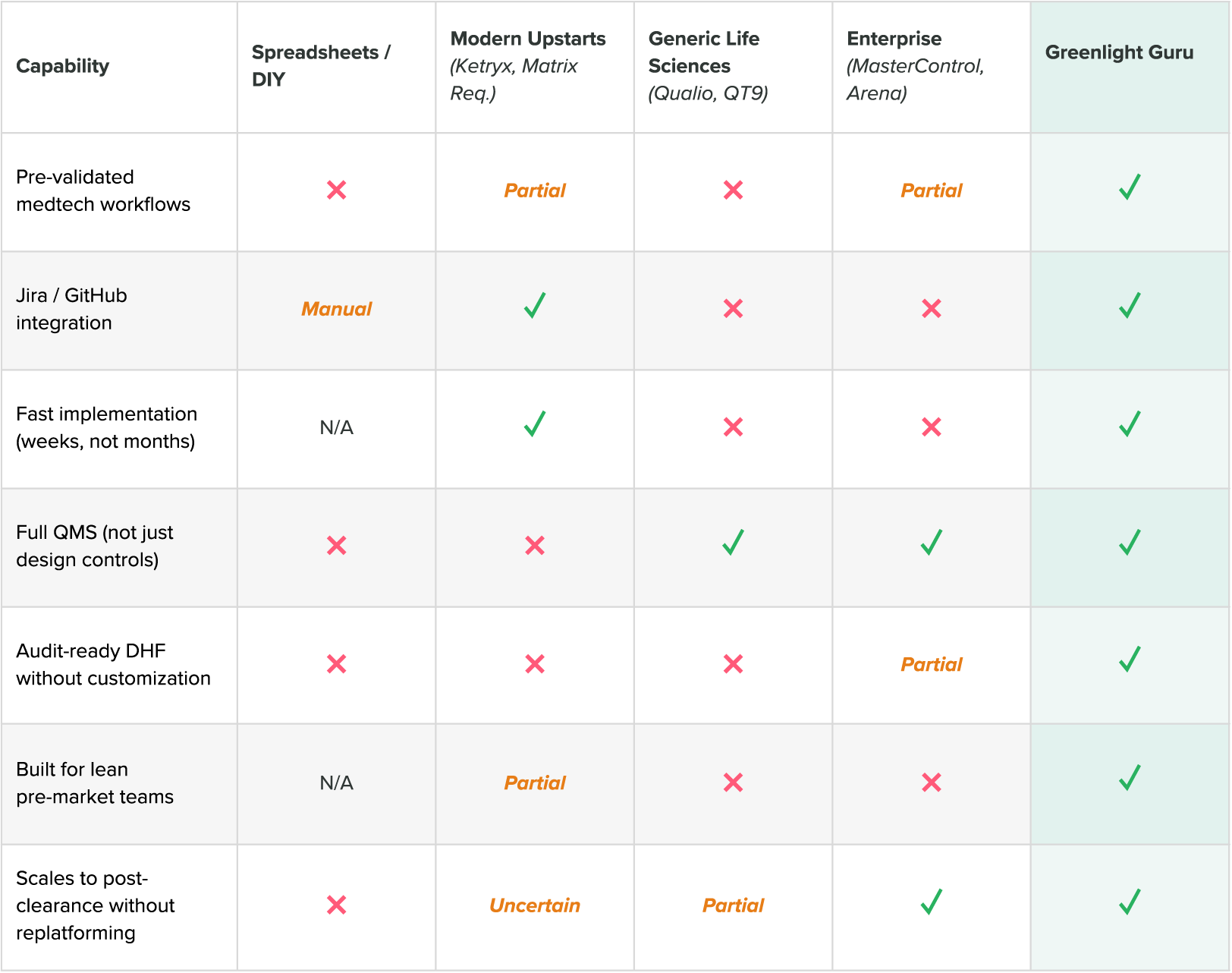
Comparing the best QMS solutions for medical devices
DIY and spreadsheets
The problem with DIY systems is that they accumulate quality debt quietly. Teams working from spreadsheets and shared drives routinely spend four to eight months in documentation cleanup before they can even begin submission prep.

Generic life sciences QMS software
The core problem is that medtech quality requirements are structurally different from pharma quality requirements. Design controls, bidirectional traceability between requirements and risk, and DHF management are not standard features in systems that weren't built around them.
Enterprise QMS platforms
Enterprise platforms are the wrong tool for pre-market teams. Implementation timelines are measured in months or years. The workflows are built for the problems of a large, commercialized manufacturer, not for a lean team trying to build a traceable DHF while simultaneously building a device.
Modern upstarts
A newer category of QMS tools has emerged, built primarily for software as a medical device (SaMD) and SiMD companies. Teams will find gaps in areas like design verification and validation documentation, supplier management, and full CAPA and nonconformance (NC) infrastructure.
Best QMS software by medtech company stage
Company Stage |
What to Prioritize in QMS Software |
Why It Matters |
Early-stage / pre-market |
Design controls, risk management, document control, traceability, training, and audit-ready records |
Your QMS foundation affects submission readiness and future scalability |
Scaling startup |
Change control, CAPA, supplier quality, audits, implementation support, and cross-functional visibility |
Quality complexity increases as teams, suppliers, and product iterations grow |
Software-first / SaMD company |
Requirements traceability, software release connections, risk, verification, validation, and developer-tool integrations |
Software teams need QMS workflows that connect engineering evidence to regulated records |
Established manufacturer |
Supplier quality, post-market quality, complaint handling, audits, CAPA, and management review |
Mature companies need connected oversight across products, suppliers, and markets |
International device company |
ISO 13485 alignment, audit readiness, supplier controls, electronic records, and global regulatory support |
International growth increases documentation, audit, and compliance complexity |
Best QMS software for different medtech companies
Best QMS solution for scaling medical device companies
With Greenlight Guru, the team at Puzzle accelerated their go-to-market timeline by 5 months in the first year of using the QMS.
Best QMS software for established companies selling devices internationally
C2DX decided to implement a purpose-built QMS from the get-go, which allowed them to pass audits with flying colors.
Best QMS software for MedTech companies to stay audit ready
MTI chose Greenlight Guru and passed six audits in one year, including ISO and MDSAP audits.
 Learn more about how we help our customers today →
Learn more about how we help our customers today →
How to evaluate QMS software vendors during a demo
Use these questions when comparing QMS software vendors:
How much ongoing engineering time does your system require after implementation, week over week?
Can quality and product teams work in the same environment without a manual handoff between them?
How long does a standard implementation take from kickoff to audit-ready?
Walk me through what happens to our traceability records when we make a late-stage design change.
How does Design and Development File assembly work?
Can you show me how a Jira ticket connects to a design requirement?
The best QMS makes quality your competitive differentiator
The best medtech companies treat quality as a competitive advantage, rather than a compliance burden. A right-sized QMS gives your team the foundation to move faster, document cleaner, and walk into every investor conversation, regulatory interaction, and partnership discussion with confidence that your records will hold.
If your team is evaluating medical device QMS software, Greenlight Guru's purpose-built QMS software for medical devices connects quality management, product development, design controls, risk, CAPA, audits, and supplier quality in one platform. See Greenlight Guru's QMS software.
Etienne Nichols is the Head of Industry Insights & Education at Greenlight Guru. As a Mechanical Engineer and Medical Device Guru, he specializes in simplifying complex ideas, teaching system integration, and connecting industry leaders. While hosting the Global Medical Device Podcast, Etienne has led over 200...
Related Posts
eQMS Buyer’s Guide: Making the Business Case for a New QMS Solution
Enterprise Quality Management System Software: What You Need To Know
11 Questions to Ask QMS Software Vendors in the Medical Device Industry
Get your free PDF
The Ultimate Guide to Comparing QMS Solutions








