What comes after launch? A guide to post-market maturity
.png?width=800&height=400&name=What%20comes%20after%20launch%20A%20guide%20to%20post-market%20maturity%20(1).png)
Getting to market is a milestone most pre-market teams have been working toward for years. Then the moment comes, all your hard work pays off, and your product finally has market access. But the work doesn't stop or settle down. Instead, it changes.
Pre-market work is milestone-driven. Every sprint, every design review, every submission step is organized around a single goal: get the device to market. The operating model is built for that, but once the device is on the market, that model stops fitting the work.
Post-market work is continuous. Complaints land in inboxes. Products need changes. Customers ask questions that turn into investigations. The regulatory bar keeps moving. And none of this comes with a handbook for how a team is supposed to do it all while also growing the business.
Launch is a shift in how the team works, not a finish line. Operating in-market requires a different way of working than getting to market, and most teams aren't warned about that in advance.
This guide exists to name the stages that teams go through once they have market access, describe what each one looks like in practice, and offer a practical path forward.
BONUS RESOURCE: Click here to download your free complaint log and form template bundle!
Table of contents
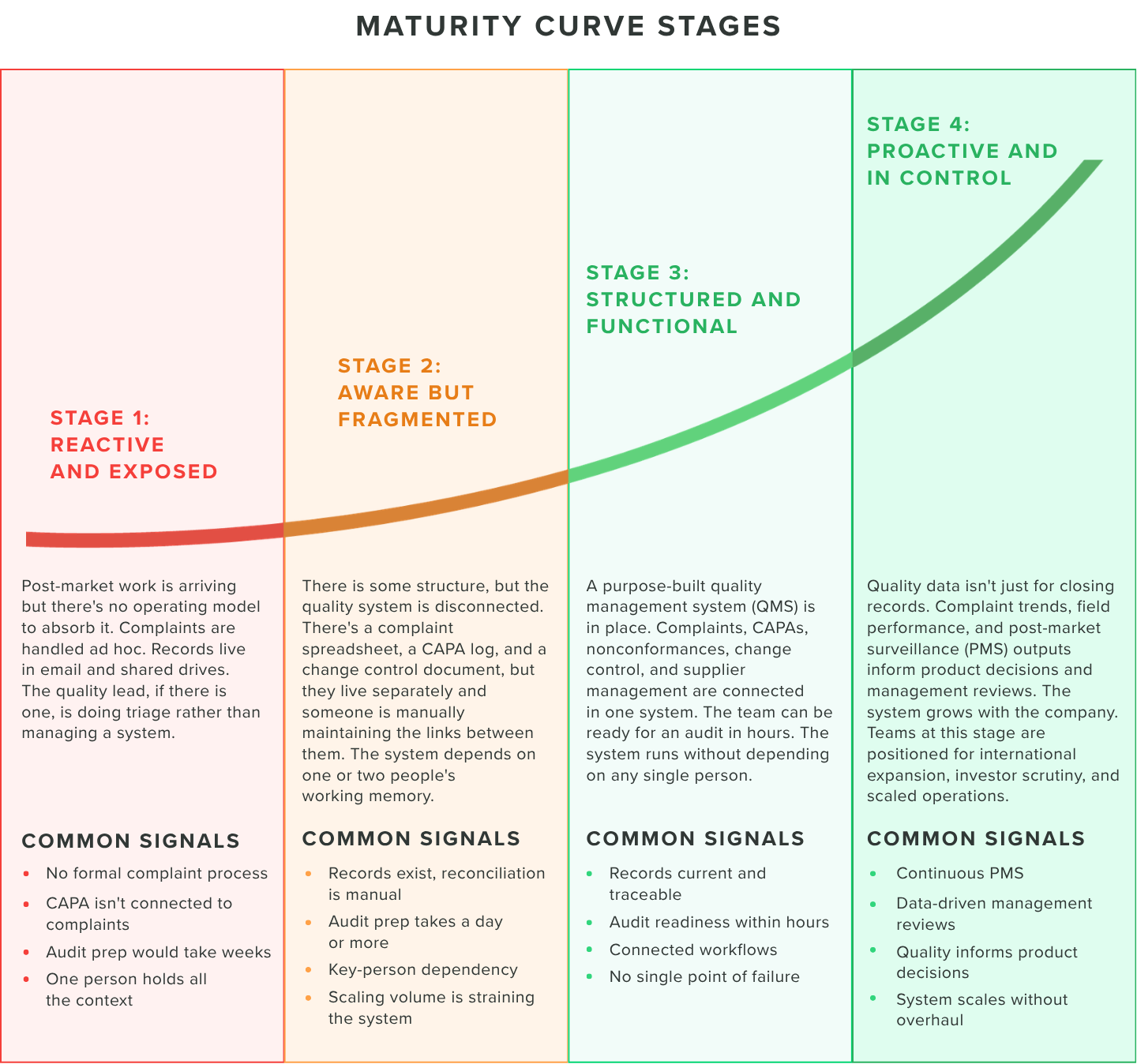
The four stages of post-market maturity
Every post-market medtech team is somewhere on this curve. The stages describe how quality systems actually operate in practice, not as a compliance benchmark, but as a day-to-day reality.
Not sure where you land? Try taking our 7 question post-market maturity quiz.
Stage 1: Reactive and exposed
This is where most teams are in their first year on the market.
What work looks like at this stage
Complaints arrive without a clear process for receiving, investigating, or closing them. The CAPA process may not exist at all, or may be informal decisions made in Slack conversations. Change control happens informally. Training is undocumented.
The real cost of Stage 1
According to published FDA annual quality system data, CAPA-related deficiencies are the single most frequently cited issue in 483 observations year after year, and complaint handling is consistently in the top three.
What's at risk under QMSR
The FDA's Quality Management System Regulation (QMSR), effective February 2026, allows inspectors to ask for documentation of management reviews, internal audits, and supplier audits, all of which used to be off-limits.
The signal that you're at Stage 1: when a complaint arrives, the first question your team asks is "where do we put this?" Not "what's our process for this?"
Stage 2: Aware but fragmented
Stage 2 teams recognize the problem and have started building structure. The challenge is that the pieces aren't connected. The team may be thinking about a solution to their problems, but inertia and a fear of an overwhelming implementation and migration are holding them back from purchasing a new eQMS.
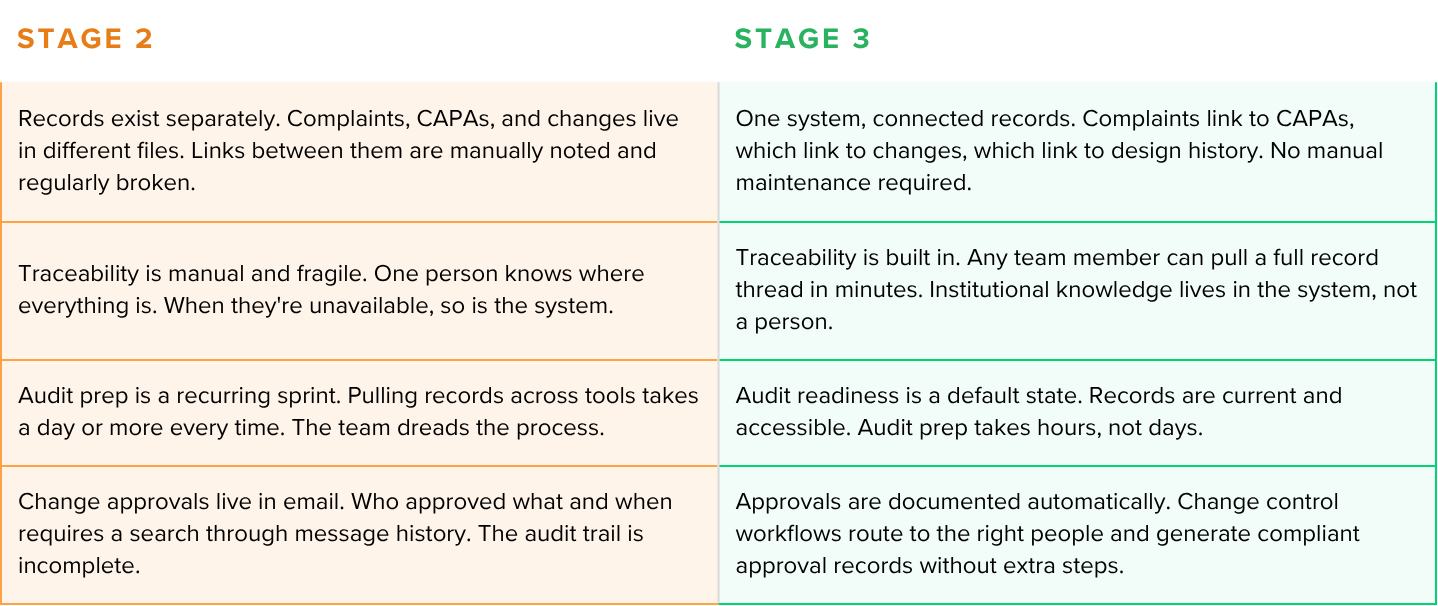
The signal that you're at Stage 2: you could pass an audit if you had two weeks to prepare. You're not confident you could pass one with two days' notice.
Stage 3: Structured and functional
Stage 3 is where post-market operations stop feeling reactive and start feeling like a system. Complaint management, CAPA, change control, supplier management, and training are connected in a purpose-built QMS.
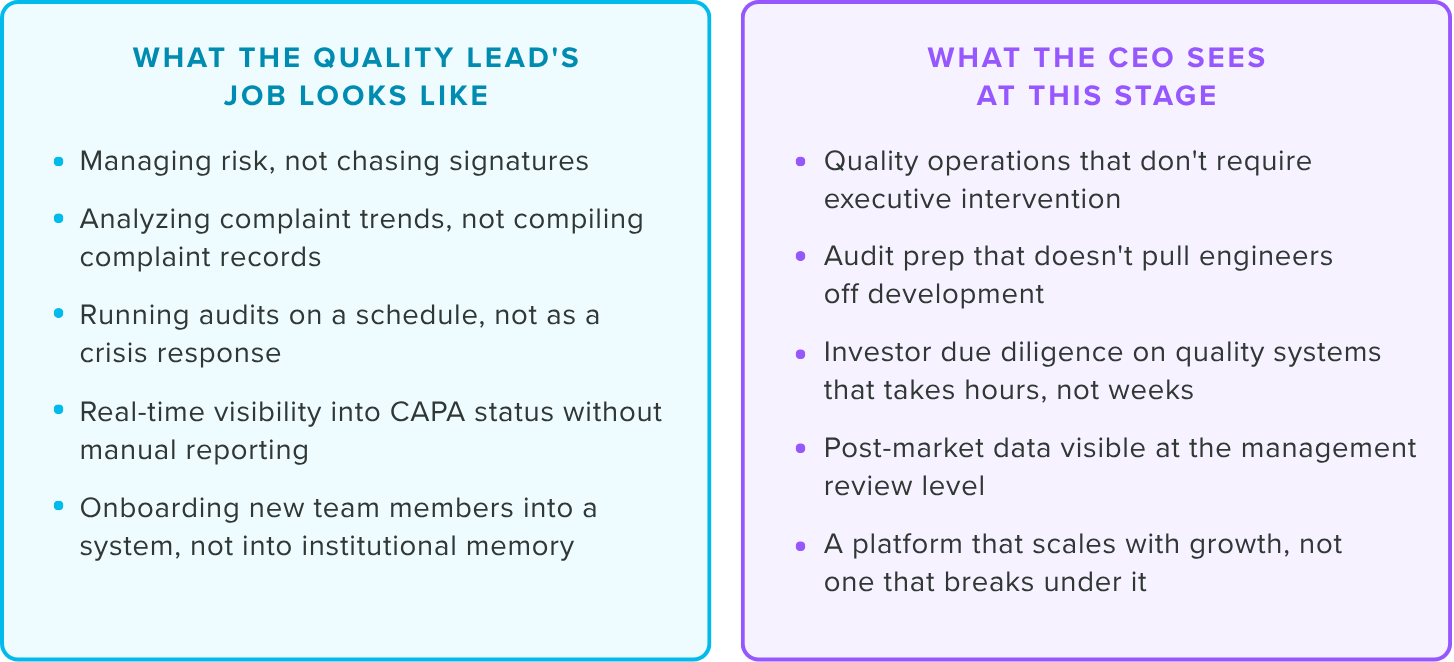
The Stage 3 ceiling
Complaints are closed, but they aren't trended. CAPAs are resolved, not analyzed for patterns.
The signal that you're at Stage 3: your records are current but you're not yet using them to drive proactive decisions.
Stage 4: Proactive and in control
In Stage 4, management reviews are built on operational metrics, complaint resolution rates, CAPA closure times, and supplier performance data, rather than on document reviews and status updates.
Why Stage 4 matters for growth-stage companies
Teams preparing for Series B funding, international market access, or a commercial acquisition will have their post-market operations scrutinized by both investors and regulatory bodies.
The signal that you're at Stage 4: you use post-market data to make product and regulatory decisions, not just to close compliance records.
How to move up the curve
Stage 1 to Stage 2: consolidate before you connect
The first move is getting complaint intake, CAPA tracking, and change control into consistent, documented formats.
Stage 2 to Stage 3: replace coordination with structure

Stage 3 to Stage 4: shift from record-keeping to signal reading

Where are you on the curve?
The 7-question maturity quiz maps your current operations to a specific stage and gives you tailored next steps. See where you land!
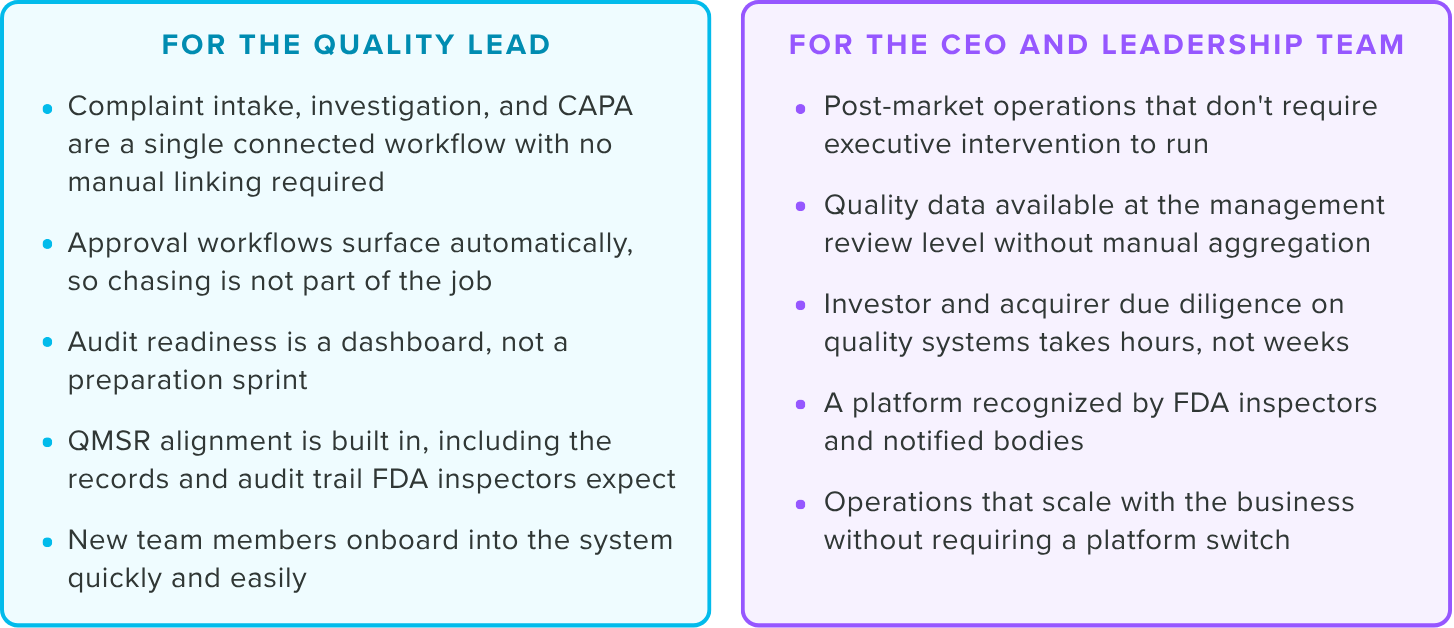
Where Greenlight Guru fits
The most common concern we hear from early post-market teams evaluating a QMS isn't cost. It's disruption.
Greenlight Guru can be configured by a single qualified person and is live in weeks, not quarters.
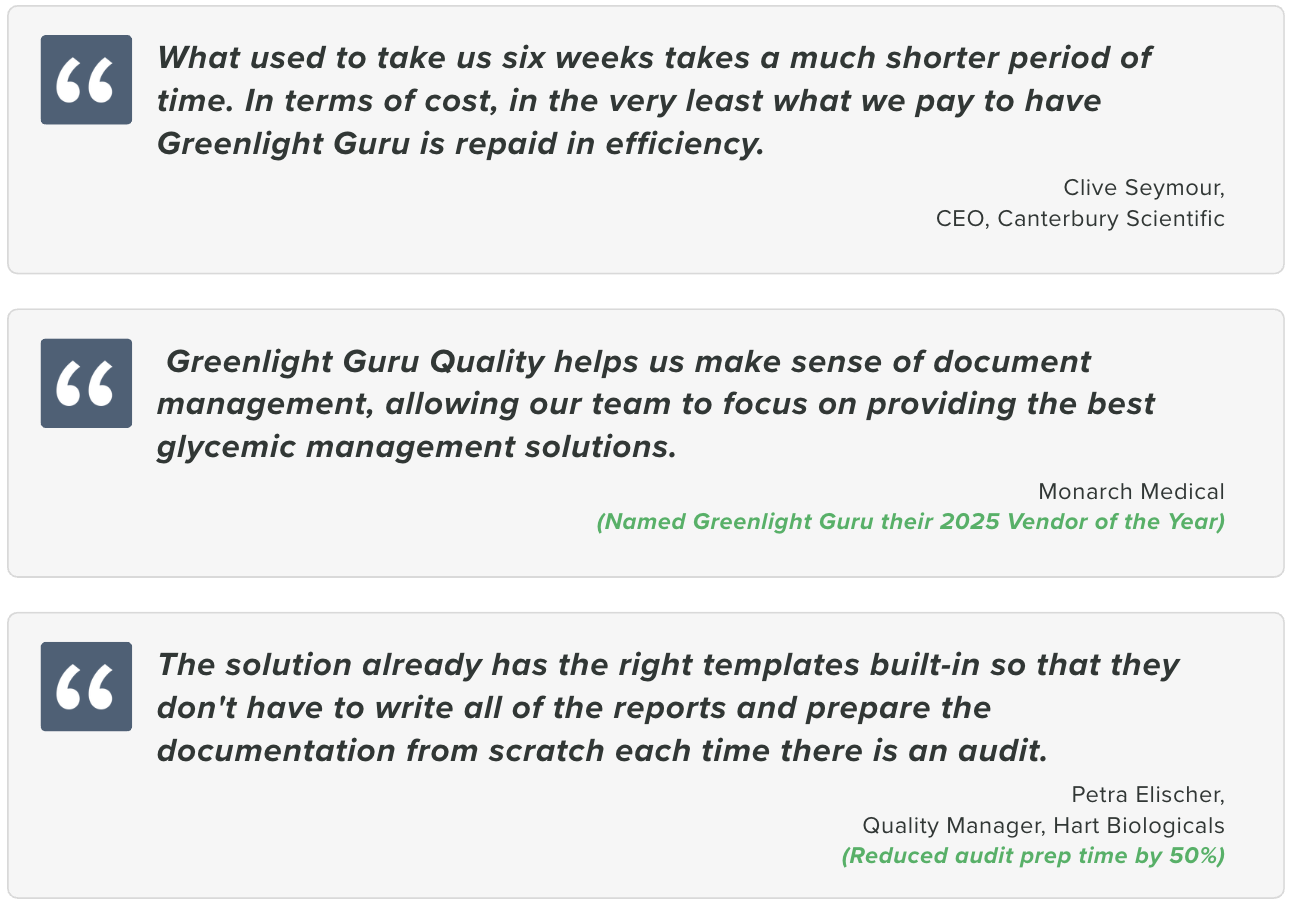
What customers say
BONUS RESOURCE: Click here to download your free complaint log and form template bundle!
Ready to move up the curve?
Most post-market teams don't struggle because they lack commitment to quality. They struggle because the tools they're using weren't built for this work.
Greenlight Guru is purpose-built for post-market medtech operations. It comes with dedicated, connected modules for complaint management, CAPA, nonconformances, change control, supplier management, and training, all structured to meet FDA QMSR, ISO 13485, and MDR requirements on day one.
If you're ready to move from reactive to in control, get your free demo of Greenlight Guru today.
Elizabeth Weddle leads product marketing at Greenlight Guru, where she focuses on understanding market shifts and customer needs - translating those insights into strategy. With more than a decade of experience driving go-to-market strategy, she helps organizations navigate change and deliver solutions that drive...
Related Posts
The Ugly Truth About Managing Design Controls on Spreadsheets
Ultimate Guide to Supplier Management for Medical Device Companies
Ultimate Guide to Postmarket Surveillance of Medical Devices
Get your free download
Complaint Log + Form Template Bundle







