Products
-
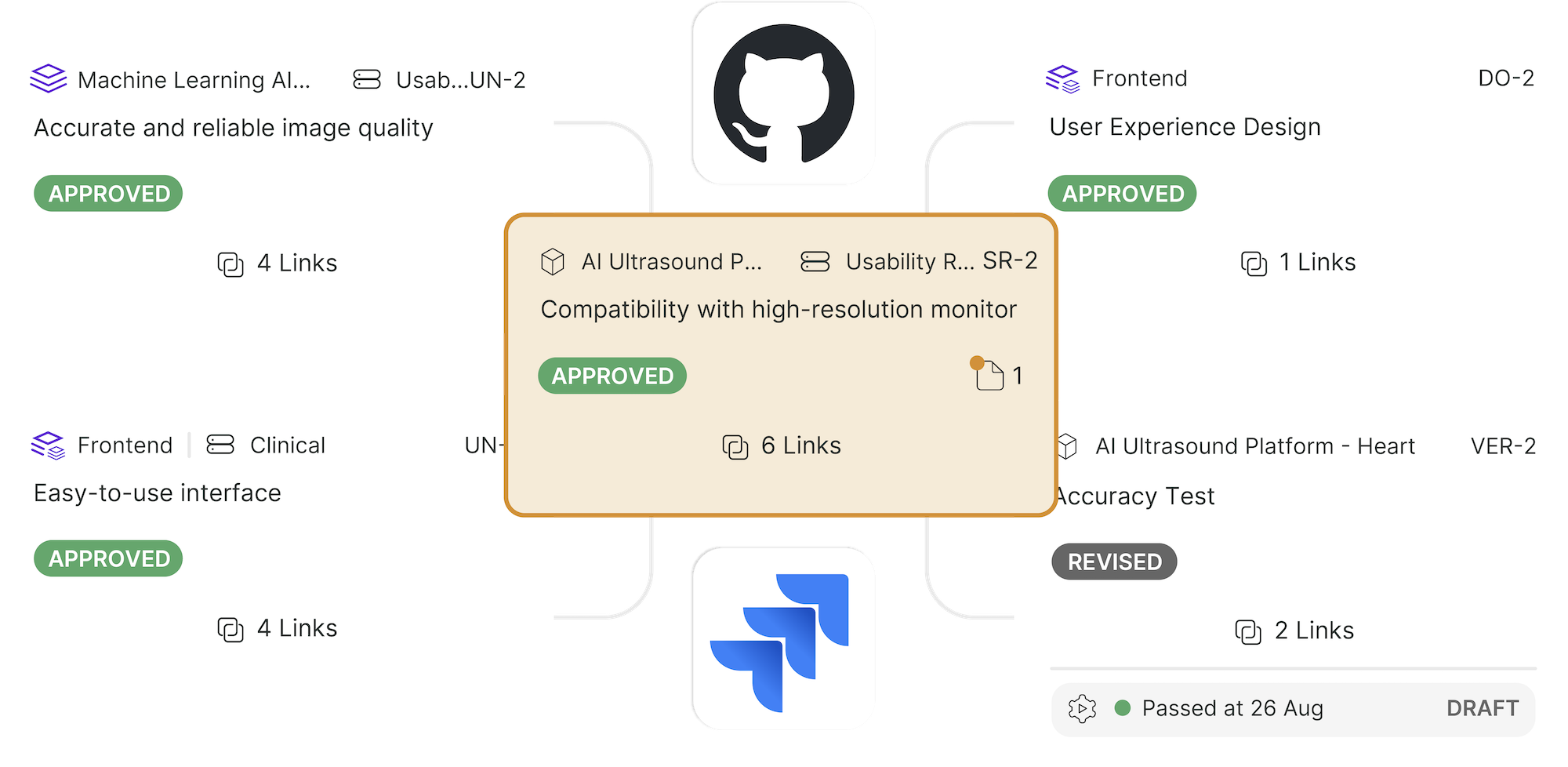
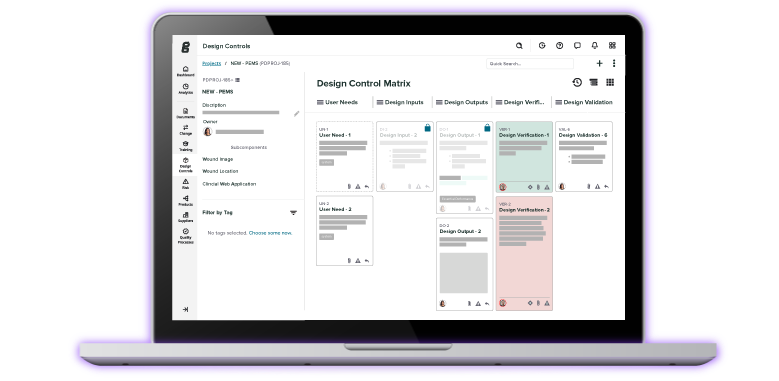
Product Development
-
Quality
-
Clinical
By Team
A modern eQMS for product-led,
fast-moving medtech innovators.
- Built to fit your process, not force it
- Stay focused on product, not paperwork
- Built for engineers and trusted by auditors
An eQMS for product-led,
fast-moving medtech innovators.
- Flexible to support how you work
- Help engineers stay compliant as they build
- Integrates with your existing tech stack
An eQMS for established and
expanding medtech teams.
- Guardrails to guide your team
- Built for global scale
- Audit-tested quality workflows
Data collection and management
designed for medtech clinical trials.
- No-code setup for any study type
- A single platform for all your studies
- Facilitating ISO 14155 and 21CFR Part 11 compliance


















.png?width=425&height=213&name=Spring%2023%20Badges%20(1).png)